Detailed introduction:
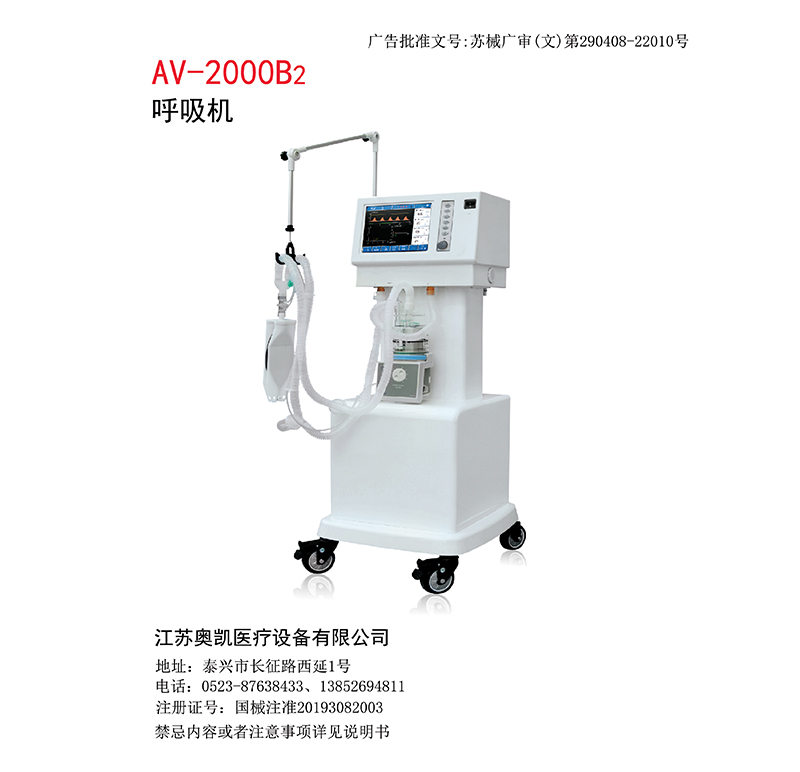
Product Name: Ventilator
Specification and model: AV-2000B2
Structure and composition: This ventilator consists of a main unit (including air circuit, electronic system, model structure, software, display), a trolley, and an air compressor (model: 40RNS, suitable for AV-2000B3 ventilator).
Scope of application: This product is used for respiratory support and respiratory diagnosis and treatment in adult patients.
Registration Certificate Number: National Medical Device Registration Approval 20193082003
Registered person information: Jiangsu Aokai Medical Equipment Co., Ltd
Production License Number: Su Shi Yao Jian Xie Production License No. 20040074
Product Technical Requirement Number: National Medical Device Approval No. 20193082003
Relative contraindications include pneumothorax and mediastinal emphysema without chest drainage, severe pulmonary hemorrhage, pulmonary bullae, hypovolemic shock, and acute myocardial infarction.



